“Doing nothing often leads to the very best of something”

I don’t think that wise sage of a bear was thinking about urine, but we can look to Winnie the Pooh for guidance in the management of subclinical bacteriuria.
Why? Let’s do a shallow dive into the issue.
Subclinical bacteriuria (called asymptomatic bacteriuria in people) is a state where bacteria are present in the bladder in the absence of disease. In the past, this has often been considered something to treat, but we now know that the bladder isn’t always a sterile environment and having bacteria in it isn’t necessarily bad. Bacteria cycle in an out of the bladder, and subclinical bacteriuria is a common state for some animals (and people).
Rarely does it need to be treated. In humans, the main indications are in pregnant women and before undergoing a urological surgical procedure. However, it’s often treated (in both people and animals), and that can lead to issues with adverse effects of antibiotics, antibiotic resistance, unnecessary cost and unnecessary hassles and stress of treatment.
When we released our first ISCAID urinary guidelines in 2011, many people pushed back at the recommendation to not treat (and not to test to look for it). By the time of our 2019 update, there was a lot less resistance, but treatment is still relatively common. As we work on the latest update, we hope for even more uptake, but realize that old habits die hard.
Subclinical bacteriuria is still often treated because of habit or fear. But, we need to focus on a few concepts.
- We treat disease, not culture results.
- Not all bugs need to die.
- If a bacterium isn’t bothering my patient, it rarely bothers me.
- Doing something isn’t always good (even though we’re hard wired to think we need to do something).
A new study provides more support to Pooh’s clinical sense.
It’s not earth shattering. It’s not surprising. However, it’s really important as more data help convince more people.
What’s the study?
It’s a study in the Journal of Veterinary Internal Medicine entitled Clinical outcomes and association with disease progression and survival of subclinical bacteriuria in cats with chronic kidney disease: a multicenter retrospective study (Le Corre et al, 2026).
They looked at 287 cats with chronic kidney disease, a population that’s at increased risk for subclinical bacteriuria. They all had urine cultures from samples collected by cystocentesis (direct sampling of the bladder through the belly wall using a needle and syringe to reduce contamination risks) and had no signs of lower urinary tract disease (e.g. straining to urinate).
- Bacteria were isolated from the urine of 38% of cats, pretty much in line with earlier estimates. Unsurprisingly, risk was higher in female cats, who were 5.3 times as likely to have bacteriuria compared to males.
- E. coli was most common, accounting for 68% of isolates, followed by Enterococcus (17%), Staphylococcus (5%) and Pseudomonas (5%). 20% of bacteria were multidrug resistant (ugh).
- 85 cats with subclinical bacteriuria got antibiotics, 41% of those before antimicrobial susceptibility data were available.
When antibiotics were given, another round of bacteriuria was identified later in 62%.
- That’s a key aspect because nothing about these cats’ susceptibility changed. We didn’t do anything to make they less susceptible to subclinical bacteriuria so it’s not surprising it happened again. It highlights the futility of treating. If we eliminate the bacterium today, odds are a new one will move in in the near future. The more we treat, the more likely we are to harm the cat with antibiotic adverse events, and the next time bacteriuria is present, a resistant one may be there. If the cat actually develops an infection that needs antibiotics, we may have limited treatment options.
When they looked at 1500 day followup, there was no association between subclinical bacteriuria and survival. That fits with an earlier study of senior cats and reams of data from humans.
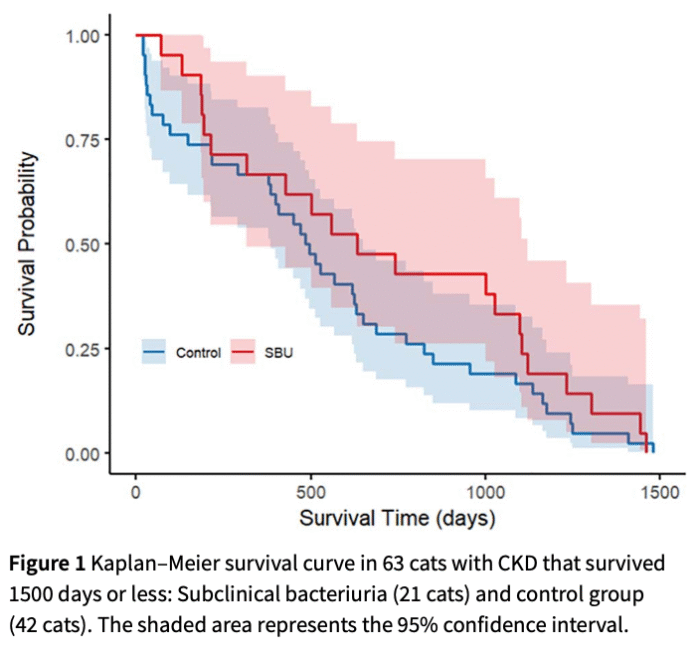
Progression of chronic kidney disease was also assessed in 134 cats. There was no difference, with progression of disease found in 42% of cats with subclinical bacteriuria and 40% of controls. The presence of multiple rounds of subclinical bacteriuria was also not associated with kidney disease progression.
There was no significant difference in development of bacterial cystitis, but there was a numerical difference that’s worth paying attention to and investigating more. That said, a question would be whether cats that got treated were more likely to have other complicating factors that were a driver for cystitis. There was also not a significant risk of pyelonephritis in untreated cats.
All this supports the notion that subclinical bacteriuria is a common and typically benign state. Treatment can sometimes eliminate bacteria, but not always, and when it does, re-infection is common. It’s possible that treatment would reduce the subsequent risk of bacterial cystitis, but even if there’s a small effect, we must consider the adverse events associated with treatment, especially treating lots of cats over and over again.
The authors conclusions?
In conclusion, we found no significant association of SBU with survival and disease progression in cats with CKD. Despite antimicrobial treatment of SBU in cats with CKD, urinary sterilization was not achieved in most cases, and progression to bacterial cystitis or pyelonephritis, although in frequent remained possible. Despite the fact that no clear guidelines for the management of SBU in cats with CKD currently exist, our results do not support routine antimicrobial treatment of this condition. Further evidence on the appropriateness of withholding treatment to manage SBU in cats with CKD remain however to be demonstrated before such recommendations can be made.
Image from https://tommccallum.com/2021/05/21/doing-nothing-often-leads-to-the-very-best-of-something/