 These days, there’s more and more doom-and-gloom information about multidrug-resistant bacteria. They’re in our hospitals, medical tourists, people on the street, our pets, our food, and pretty much anywhere else you can think of. We can now add crow poop to the list too.
These days, there’s more and more doom-and-gloom information about multidrug-resistant bacteria. They’re in our hospitals, medical tourists, people on the street, our pets, our food, and pretty much anywhere else you can think of. We can now add crow poop to the list too.
It’s almost to be expected, really. We know that birds can carry various resistant bacteria, and the more contact birds have with human environments and food animal environments, the greater the chance these bacteria are going to be transmitted between them (in one direction or another). It’s important to remember that resistant bacteria are also present in nature, independent of human activities.
A recent report of a pretty high profile multidrug-resistant bacterium – vancomycin-resistant Enterococcus (VRE) – in birds wasn’t all that surprising. The study (Oravcova et al, Environmental Microbiology 2013) reported finding enterococci carrying the vanA resistance gene in 2.5% of 590 crows sampled in multiple US states. It was quite interesting though, because VRE is (in North America) a human-associated bacterium. It’s a little more muddled in Europe where VRE was an issue in food animals, in part due to former use of a drug related to vancomycin (avoparacin) in some food animal species. Here though, we rarely see VRE in anything species but humans. This raises some interesting questions about where these crows picked up VRE, if they are able to carry the bacterium for long periods of time, and if they can act as a source of human or animal infection.
Does this bother me? No. It’s of academic interest, but not something that’s going to pose a real risk to me. I tend not to walk under trees full of crows with my mouth open, and I’m pretty sure I’d wash my hands if a crow pooped on them. Yes, there’s the chance that I could have unnoticed contact with contaminated crow poop remnants on an outdoor surface, but the odds of it containing viable VRE are pretty low, and there are lots of other things that I’m more likely to pick up in my daily activities. In terms of VRE, I’m presumably more likely to be exposed in other ways than from crows. However, the study is still important in that it shows how widespread antimicrobial resistance is, how complex the issue is and how we need to do more to understand the ecology and epidemiology of various resistant bugs.
There’s no need to go exterminating crows, but Johnny Depp may want to consider an alternative style of hat.
 Last week, I mentioned the
Last week, I mentioned the  Pet aquatic turtles have been implicated in three outbreaks of salmonellosis involving 43 US states over the past year and a half. Disappointing, but not surprising.
Pet aquatic turtles have been implicated in three outbreaks of salmonellosis involving 43 US states over the past year and a half. Disappointing, but not surprising. A little knowledge can be a bad thing. We see that with zoonotic diseases. Awareness is great. However, a little bit of awareness can be a problem if it’s enough make people paranoid but not enough to help them understand the real risks. This can lead to excessive and illogical responses (often ending with "…get rid of the cat").
A little knowledge can be a bad thing. We see that with zoonotic diseases. Awareness is great. However, a little bit of awareness can be a problem if it’s enough make people paranoid but not enough to help them understand the real risks. This can lead to excessive and illogical responses (often ending with "…get rid of the cat"). I’m getting ready for next week’s
I’m getting ready for next week’s  When it comes to hand hygiene, there is an unfortunately all-too-common misconception that wearing gloves makes hand washing or using alcohol-based hand rub unnecessary. In veterinary and human medicine, gloves, like hand hygiene, are typically used for two reasons: to prevent spread of germs or chemicals from a patient/person/object/surface to a person’s hands, and/or to prevent the spread of germs or chemicals from a person’s hands to a patient/person/object/surface. However, gloves are not the infallible barrier to germs that many people would like to think they are. Here are a few reasons why:
When it comes to hand hygiene, there is an unfortunately all-too-common misconception that wearing gloves makes hand washing or using alcohol-based hand rub unnecessary. In veterinary and human medicine, gloves, like hand hygiene, are typically used for two reasons: to prevent spread of germs or chemicals from a patient/person/object/surface to a person’s hands, and/or to prevent the spread of germs or chemicals from a person’s hands to a patient/person/object/surface. However, gloves are not the infallible barrier to germs that many people would like to think they are. Here are a few reasons why: In my line of work, I deal with feces from a lot of different species. Never once, however, have I thought to model defecation pressure and distances.
In my line of work, I deal with feces from a lot of different species. Never once, however, have I thought to model defecation pressure and distances.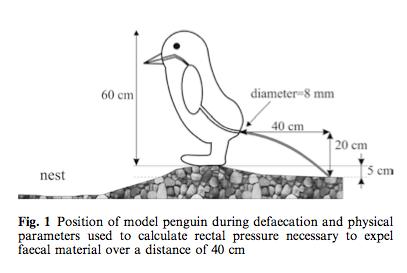 I won’t get into all the of the calculations. Suffice it to say, the image at right will probably not been seen in too many other scientific papers.
I won’t get into all the of the calculations. Suffice it to say, the image at right will probably not been seen in too many other scientific papers. A few days ago, I received a question about whether bathing a young child and a dog together in the bathtub is a bad idea.
A few days ago, I received a question about whether bathing a young child and a dog together in the bathtub is a bad idea. A local county newspaper had a
A local county newspaper had a 