Antimicrobials are often used at the time of surgery, but it’s widely accepted that there is tremendous overuse of antimicrobials in this context in both human and veterinary medicine. Antimicrobial prophylaxis is indicated in some surgical patients to reduce the risk of surgical site infection, but in a large percentage of cases use of antimicrobials is actually unnecessary and is based more on habit or fear (i.e. more to make the surgeon or pet owner feel better, versus actually helping the patient).
Clinical guidelines are an advancement in care, and the field of antimicrobial guideline development has progressed significantly in recent years. We’ve moved from primarily expert-opinion-based guidelines to evidence-based, structured guideline development, which should yield stronger, less biased and more defensible guidance, but guidelines are never perfect, since there are typically still lots of evidence gaps. Also, guidelines are meant to cover the majority of situations, not every possible case, so there are always exceptions to the “rules.” Nonetheless, good guidelines help support good clinical decisions.
The European Network Optimization of Veterinary Antimicrobial Therapy (ENOVAT) has just released their new 2025 guidelines for surgical antimicrobial prophylaxis in dogs and cats. These guidelines are the culmination of several years of work, and are underpinned by a thorough scoping review of antimicrobial prophylaxis in companion animal surgery (Sorensen et al. 2024) and a systematic review on the same topic (coming soon).
The guidelines have a heavy emphasis on when NOT to use antimicrobials, since that’s what the evidence supports, but they also highlight situations where antimicrobials are recommended, and they provide details about optimal approaches for when they’re needed.
The guidelines used a GRADE-based approach, which culminates in a strong or conditional recommendation for or against each intervention (or a conclusion that we can’t make a recommendation either way).
For strong recommendations, the guidelines say “we recommend…”
- Strong recommendations for an intervention (i.e. antimicrobial prophylaxis) are made based on moderate to high certainty evidence of the effect of the intervention, plus supporting value amongst various other domains (e.g. importance of the problem, benefits, harms, cost:benefit, equity, acceptability).
- Strong recommendations against an intervention can be made based on similarly moderate-high certainty evidence or where there is lower certainty evidence about the effect but moderate/high certainty evidence about potential harms. The default is not to use the intervention, so a strong recommendation against can be made without solid data showing it doesn’t work.
For conditional recommendation, the guidelines say “we suggest…”
- Conditional recommendations are made when the recommendation is based on low certainty evidence or when there is a lot of uncertainty or variability about acceptability, applicability, equity or other factors that indicate it might not be an ideal or preferred approach for most. (For example, we’re not going to make a strong recommendation for a treatment that’s so expensive only a small subset of the population could ever use it.)
- A conditional recommendation doesn’t mean that the treatment is less effective than one with a strong recommendation, it simply means the confidence and certainty behind the recommendation is lower, and/or that its value is more situational, being a preferred choice in some situations but not others.
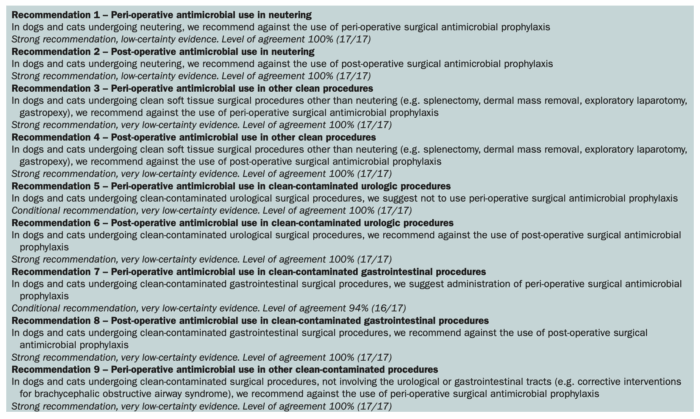
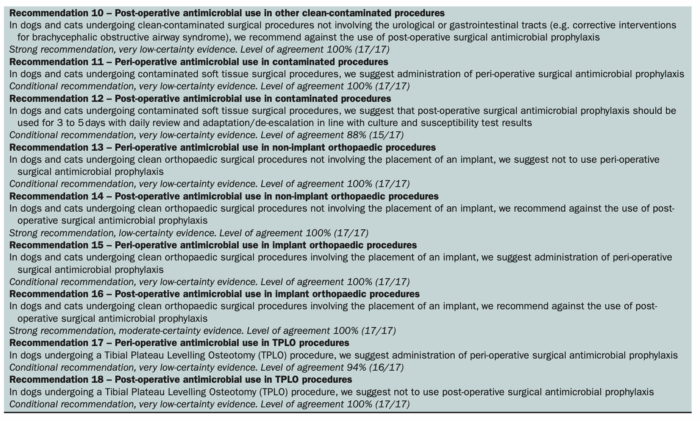
Below is a quick synopsis of the recommendations (in tiny print). Check out the full text article (which is open access) for more details on each recommendation and the evidence behind them all.