Tularemia is a nasty bacterial disease. The bug that causes it, Francisella tularensis, is a category A bioterrorism agent (along with things like anthrax, botulism, plague, smallpox and Ebola virus). It’s classified as that because it’s highly transmissible and causes serious disease, so it’s something you definitely don’t want.
Tularemia is a nasty bacterial disease. The bug that causes it, Francisella tularensis, is a category A bioterrorism agent (along with things like anthrax, botulism, plague, smallpox and Ebola virus). It’s classified as that because it’s highly transmissible and causes serious disease, so it’s something you definitely don’t want.
The bacterium circulates in the wild, most often associated with rabbits and rodents. Human infections are usually associated with exposure to these types of wild animals (including things like – gross as it is – running over a wild rabbit with a lawnmower and aerosolizing the bacterium). However, one complicating factor with this bug is that it can be spread a lot of different ways, including via tick and fly bites, water and minor contact with infected animals (e.g. just touching their skin).
Among domestic pets, cats are most commonly implicated in human infection, because cats with outdoor access tend to hunt species that harbor tularemia (fitting with the “it’s better off for cats to be indoors if they tolerate it” statement I made in my last post… that got lots of comments both for and against), and because they are fairly susceptible to infection when they get exposed. However, a variety of animals can harbor and transmit the bacterium.
Dogs are lower risk but not no risk, as a recent paper in Zoonoses and Public Health (Kwit et al. 2018) explains. They looked at US tularemia surveillance data from 2006-2016, and focused on the 24 cases that were deemed dog-associated. Whether those were all truly from dogs can’t be guaranteed, and it’s possible that some of the other cases were dog-associated but the link wasn’t investigated or identified.
Among the highlights:
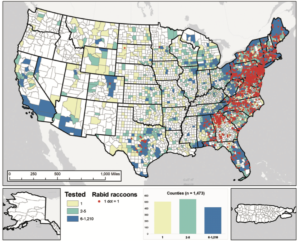
- Most cases were from Missouri, Colorado and Kansas, consistent with what we know about tularemia in general in the US.
- Outcome was known for 20 of the affected people. Two died.
- 50% of cases were thought to be from direct contact with dogs, including bites or scratches, but also contacts such “face snuggling” and licking
- 33% of people were exposed from contact with carcasses brought home by their dogs.
- 17% of cases were associated with ticks thought to have been brought in by dogs. Implicating dogs as a “source” of ticks in a household is tough, because if a dog is exposed to ticks, people in the same area have a similar risk of being exposed if they’re outside. The bigger risk might be an increased tendency to spend time in tick-infested areas when walking said dogs.
- Not much dog-health information was reported. Four people had contact with “ill” dogs but it’s not clear how many dogs might have had tularemia vs been infected without signs of disease vs been contaminated by the bacterium but not infected (and been “ill” from something else entirely).
- One dog was diagnosed with tularemia. That’s not too surprising since dogs tend not to get sick or at least not get very sick from tularemia (as opposed to cats), so it’s easy to miss a potential dog case.
The take-home messages are pretty straightforward. Tularemia is very rare and very low of my list of disease concerns when it comes to dogs. However, avoiding tularemia involves common sense practices that should help reduce a lot of other risks at the same time.
- Reduce roaming of dogs, especially dogs that are able to or like to interact (e.g. chase, hunt) wildlife like rabbits.
- If your dog brings back a carcass, handle it with care. Try to avoid touching it and, if you have to, wear gloves, then wash your hands after removing the gloves. Avoid contact with the dog’s mouth for a while (who knows how long… maybe a couple hours?) if it had a carcass in its mouth.
- Reduce tick exposure as much as possible (for dogs and people).
- Use tick preventives in dogs to reduce the risk of tickborne disease exposure. A variety of ticks can transmit F. tularensis, including the dog tick (Dermacentor variabilis), the wood tick (Dermacentor andersonii) and the Lone Star tick (Amblyomma americanum).
- Mention rabbit and rodent exposure potential to your veterinarian if your dog gets sick.
- Mention your dog ownership, your dog’s potential contact with wildlife, and any contact you have had with wildlife (dead or alive) to your physician if you get sick.